Our group’s research includes a diverse array of projects within the field of chemical biology with natural products as the focus.
There are 4 main complementary and overlapping research projects ongoing within our group:
- Biosynthetic Elucidation: We are particularly intrigued by medicinally relevant and biosynthetically exotic natural products, and in determining the chemistry, enzymology and genetic programming that underpins their assembly.
- Harnessing Biosynthesis To Generate New to Nature Natural ProductsNatural products, compounds generated by living organisms, play a critical role in medicine. Over 60% of anticancer agents and over 70% of antibiotics are based on natural products. The generation of analogues of these compounds is critical in order to probe the identity and nature of the targets, determine a compound’s mechanism of action, explore structure activity relationships and develop designer compounds with improved activities an bioavailability.

By blending together synthetic biology and synthetic chemistry we are able to expeditiously access analogues of structurally complex natural products which would other wise be hard to access using synthesis alone.
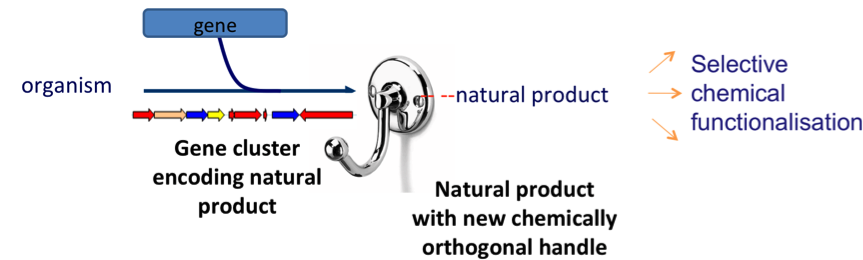
Our approaches to analogue generation include precursor-directed biosynthesis, mutasynthesis, semi-synthesis, total synthesis and a new approach that we have pioneered called GenoChemetics.
GenoChemetics– a new paradigm in natural product analogue generation
In this powerful approach, a gene, encoding a reactive and selectively chemically modifiable handle, is introduced into an organism to act in concert with an existing biosynthetic pathway. By developing mild reaction methodologies, we can selectively derivitise the natural product without the need to employ protecting group strategies.
Gene Insertion enables Selective Chemical Modification
We have developed a new approach to enable the manipulation of biosynthetic pathways. By introducing a gene to act in concert with an existing biosynthetic pathway we have installed a handle enabling selective chemical modification of the natural product.
Modification at this handle affords the potential to selectively ligate or immobilise the unnatural product. We plan to extend this concept further by genetically introducing other chemical handles.
- Natural Product Discovery (with a particular focus on finding new antibiotics with novel scaffolds and clinically unexploited modes of action) – we employ state-of-the-art approaches and discovery is directed by combining genome reading, metabolomics and bioactivity assessment.
We look in environmentally unusual niches in order to find unique chemistries and enzymologies.
- Discovering and Developing Enzymes for Use in Synthesis
(Including our engineered plug- and-play biocatalytic biofilm platform for use in flow chemistry)
Developing biotransformations for organic synthesis
Oftentimes the employment of an enzyme as part of a synthesis can significantly aid efficiency.
A key interest within our group is the development of biotransformations that may be readily uptaken by main-stream organic chemists who would normally be reluctant to employ enzymatic approaches. Our convenient approach to generating tryptophan analogues is being gradually embraced by a number of major synthetic chemistry groups across the world including the Ley group in Cambridge.
We are developing simple biotransformations to enable access to a variety of unnatural amino acids and to nucleoside analogues (In funded collaboration with GSK, Stevenage). Our work on discovering and developing new unusual halogenases as tools for bioctalysis and synthetic biology has attracted funded collaboration with AZ and Syngenta.
In complement to our biocatalyst discovery and development programmes we have developed a method of engineering biofilms of recombinant microorganisms in order to make robust biocatalysts of as potential components of flow chemistry. The engineered biofilm is considerably more robust and catalytically active than the immobilised enzyme or the natural biofilm (fig. 8). This platform has the potential to be used as a plug and play system into which genes encoding a biotransformation of choice may be introduced. Excitingly, we have recently demonstrated that the enzyme catalyst is rapidly and completely regenerated within the biocatalyic biofilm, affording biocatalytic longevity and the tantalising prospect of using gene switching to change the biofilms catalytic function.


Figure 2 Top Left: natural biofilm after 6 days, Bottom Left: significantly more mature and more stable engineered biofilm after 6 days. Right Hand Side: conversion of haloindole to halotryptophan; reaction profile for (i) 5-fluoroindole (biofilm) (ii a) 5-chloroindole (biofilm), (ii b) 5-chloroindole (immobilised enzyme), (ii c) 5-chloroindole (incubated with twice the biomass of free cells) (iii) 5-bromoindole (biofilm).
